Chapotera cheNhangande Dzakadevana
| Nhangande ye Mishonga | Udorongodzwa hwe nhangande dze mushonga
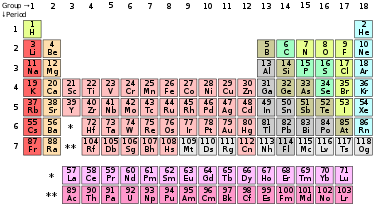
Tafura ye Rupotera (kureva periodic table; periodic table of elements) kana Chapotera cheNhangande Dzakadevana chinyorwa chinotaridza Nhangande dzemishonga dzakadungamidzana pamindanda. Apa Nhangande dzinenge dzakarongwa dzichichitevedza zvipimwa zvakacherechedzwa nenyanzvi kuti zvinoramba zvichidzokorora. Chapotera chinotaridza Nhangande dzakatevedzana: nehukuru hwenhamba yeHatomu; kurongeka kwegute rema electron nedzimwewo faniro dzakacherechedzwa. Nhangande dzinonyorwa dzichimirirwa nemavara anodudza mazita acho, imwe neimwe mubhokisi rayo.
Kupiwa kwezita
[chinjirudza | chinjirudza mabviro]Shoko rokuti kudevanisa rinoreva kuronga wakadungamidza zvichitevedza hungava: hukuru; huremu; hwasha; zera kana chimwe chingacherechedzwa chinokwaniswa kupimiwa. Sokutaura kwaitwa pamusoro pane zviripo zvakacherechedzwa paNhangande zvinotigonesa kudzidevanisa saizvozvi dzakaita ruwarikano rwemindanda.
Rupoterera zvinenge zvoturikira periodicity - apa kureva kupota kunoramba kuchidzoka panhambo inozivikanwa.
Boterero (Period) apa kureva mizera yakachinjika paChapotera. Sokuti muzera unovambira pane Hydrogen (H) kusvika pane Helium (He) iboterero rokutanga. Boterero rechipiri rinovambira pane Lithium (Li) kusvika pane Argon (Ar).
Boka (Group) apa kureva shongwe dzinodzika kubva kumusoro ichienda zasi paChapotera. Sokuti shongwe inovambira pane Beryllium (Be) kusvika pane Radium (Ra) iboka rechipiri. Boterero regumi rinovambira pane Nickel (Ni) kusvika pane Darmstadtium (Ds) .
Nhangande Dzakadarikira (transition elements) imhando yenhangande dzine chikokerero (shell/orbital) chezasi chisina kuzara asi chikokerero chekumucheto chizere nema electron anokwanamo. Nhangande rudarikira dzine nhamba dzeHatomu dzinoti: 21–29, 39–47, 57–79, uye 89–107. Saka zita rokuti Nhangande Dzakadarikira rinobva pakuti dendera remukati igasva asi dendera rekumuchete rizere. Nickel imhando yenhangande rudarikira.
Tafura
[chinjirudza | chinjirudza mabviro]| Error: No name provided | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Group | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | ||||||||||||
| Alkali metals | Alkaline earth metals | Pnictogens | Chalcogens | Halogens | Noble gases | |||||||||||||||||||||||||
| Period |
||||||||||||||||||||||||||||||
| 2 | ||||||||||||||||||||||||||||||
| 3 | ||||||||||||||||||||||||||||||
| 4 | ||||||||||||||||||||||||||||||
| 5 | ||||||||||||||||||||||||||||||
| 6 | ||||||||||||||||||||||||||||||
| 7 | ||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||
